How do we diagnose dementia and what could improve this process?
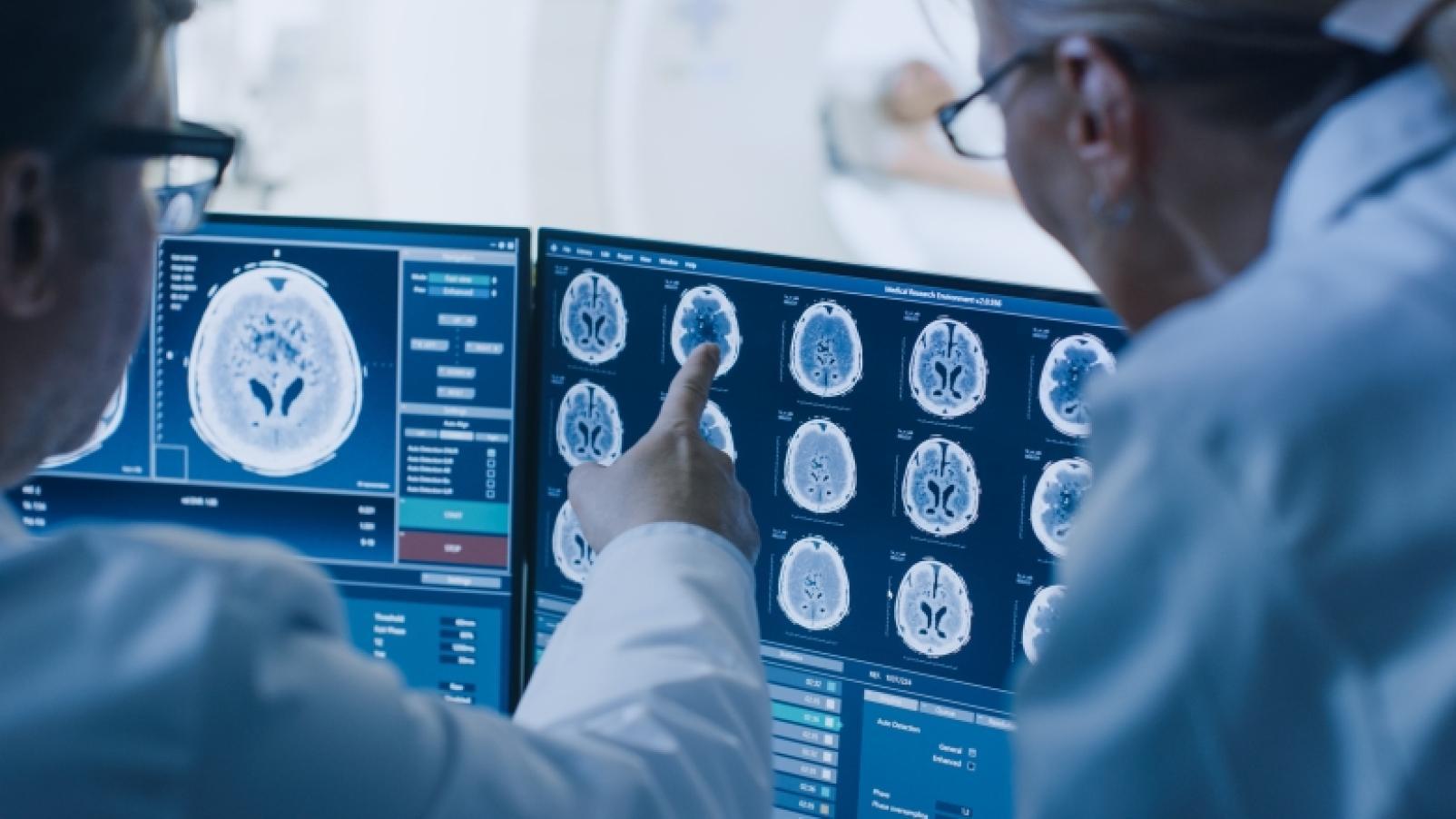
What is involved in the diagnosis process?
If you are concerned about changes in your thinking or memory or that of a loved one, consulting a General Practitioner (GP) at an early stage is critical. They may refer you on to a specialist or a memory clinic for further investigations. An assessment might include:
Medical history - the GP will ask about past and current medical problems, family medical history, any medications being taken, and the problems with memory, thinking or behaviour which are causing concern. They may also ask to hear from your partner, a close friend or family about what they have noticed.
Physical examination – Diagnosing dementia is done by a process of elimination and therefore, this may include tests of the senses and movement function, as well as heart and lung function, to help rule out other conditions.
Laboratory tests – will include a variety of blood and urine tests to identify any possible illness which could be responsible for the symptoms.
Neuropsychological or cognitive testing – a variety of tests are used to assess thinking abilities including memory, language, attention and problem solving. This can help identify specific problem areas, which in turn helps identify the underlying cause or the type of dementia.
Brain imaging – there are certain scans that look at the structure of the brain and are used to rule out a range of conditions including brain tumours or blood clots in the brain as the reason for symptoms, and to detect patterns of brain tissue loss that can differentiate between different types of dementia. Other scans look at how active certain parts of the brain are and can also help discriminate the type of dementia.
Psychiatric assessment – to identify treatable disorders such as depression, and to manage any psychiatric symptoms such as anxiety or delusions which may occur alongside dementia.
What does the future of diagnosis look like?
For Dementia Australia Honorary Medical Advisor Associate Professor Michael Woodward AM, the best diagnosis is one that is made without delay and uses the latest diagnostic techniques.
“Amyloid PET scans and Cerebrospinal Fluid are the gold standard for diagnosing Alzheimer’s disease,” Associate Professor Woodward said.
“There are also blood tests being developed that will hopefully be available soon as diagnostic tools. These tests are not quite at the stage where we can use them widely, but we are getting close. These blood tests being developed look to be quite accurate when used by a specialist.”
As well as blood tests, researchers are working on a way to use eye scans to identify people at risk of developing dementia.
In 2013, the Dementia Australia Research Foundation awarded a Postdoctoral Fellowship to Associate Professor Mojtaba Golzan to study if small changes in the blood vessels in the eye may provide an early indication of Alzheimer’s disease.
Since then, Associate Professor Golzan and his team at the University of Sydney have identified a range of biomarkers in eye scans which could indicate the onset of Alzheimer’s disease, as well a portable device, powered by artificial intelligence, that can detect changes in these biomarkers.
“Because it is a low cost, and non-invasive, we believe this test could be a very valuable screening tool to identify people who are at risk of developing dementia to be referred for further testing,” he said.
Associate Professor Golzan said the eye test also has the potential to track the progression.
“We still need to go through clinical trials and get regulatory approval for this test, which will take some time. Optimistically, this test may be available for use in around five years, but that is assuming everything goes right. Realistically, it could be a bit longer than that until this test is available for widespread adoption,” he said.
Alzheimer’s disease is one of the most common forms of dementia, other types have different diagnostic treatment. For more information about this process, visit our Diagnosing dementia help sheet.
What can support a timely and accurate diagnosis?
As well as using the appropriate tools and ensuring diagnosis is timely, Associate Professor Woodward said it is important for clinicians to form good relationships with those they are diagnosing.
“It is important for clinicians to take into account the fears and the expectations of patients and their families. Every patient and every family is different,” Associate Professor Woodward said.
Adrian was diagnosed with younger onset dementia in 2020 and had a very good relationship with his clinician. This helped made his diagnosis journey a relatively smooth one.
“My diagnosis journey started when I went to my doctor, who I had been seeing for 25 years, to get a small cyst on my neck removed. After cutting it off, he then very gently asked me how I was going,” Adrian said.
“My mother and father had both recently died, so I told him I was dealing with that and he very gently said to me that maybe I should go and see a psychiatrist.
"I had also raised concerns about memory loss with my GP and he referred me to a psychologist for an neuropsychological assessment.
"The neuro-psychologist confirmed that my level of functioning appeared low and referred me for an MRI."
Not long later, Adrian was diagnosed with younger onset dementia.
“I was lucky enough to have quite a good diagnosis journey. I know that getting diagnosed can be difficult and stressful, but it’s also so important,” Adrian said.
Where do I start?
If you are worried about your cognition or that of a loved one the best place to start is at the doctor. You can also contact the National Dementia Helpline 24 hours a day, seven days a week, 365 days a year on 1800 100 500. We are here to help.
You can also track and optimise your brain health over time with the new free BrainTrack app. BrainTrack helps individuals explore common cognitive concerns and provides brain health information through fun, travel-themed games.
Want to read more about diagnosis? Check out these articles:
Is there a point to getting a diagnosis of dementia? Many people can live well with dementia for many years but as there is no cure is there any point to getting a diagnosis?
Now I have a diagnosis, what’s next? People first diagnosed are often unsure of how to feel or where to turn. In this article we hear from people living with dementia about what they experienced, and what everyone can do to support people diagnosed with dementia.
Can a diagnosis of dementia inspire a positive outlook? When Russ started having trouble with his vision he put it down to his optical prescription and was initially misdiagnosed. Russ says he still hates the day they told him he had dementia. Find out how he changed his outlook.
Want to read more stories like this one? Subscribe to Dementia Australia’s eNews.
